At TRIMTECH we are developing a unique pipeline of protein aggregate selective degraders known as TRIMTAC® and TRIMGLUE® for the treatment of severe neurodegenerative and inflammatory diseases.
Our unique approach directs the innate properties of TRIM21, a novel E3 ligase that is expressed in most tissues, to potently and selectively degrade disease-causing protein aggregates, while preserving healthy native versions of these proteins to maintain proper cell function.
Transforming the proteinopathy medicine landscape
TRIMTECH’s technology overcomes the limitations of genomic modalities and antibody therapeutics as well as current TPD approaches by delivering selective and rapid degradation of targets that underpin neurodegeneration and inflammation.
Harnessing TRIM21’s unique mechanism of action
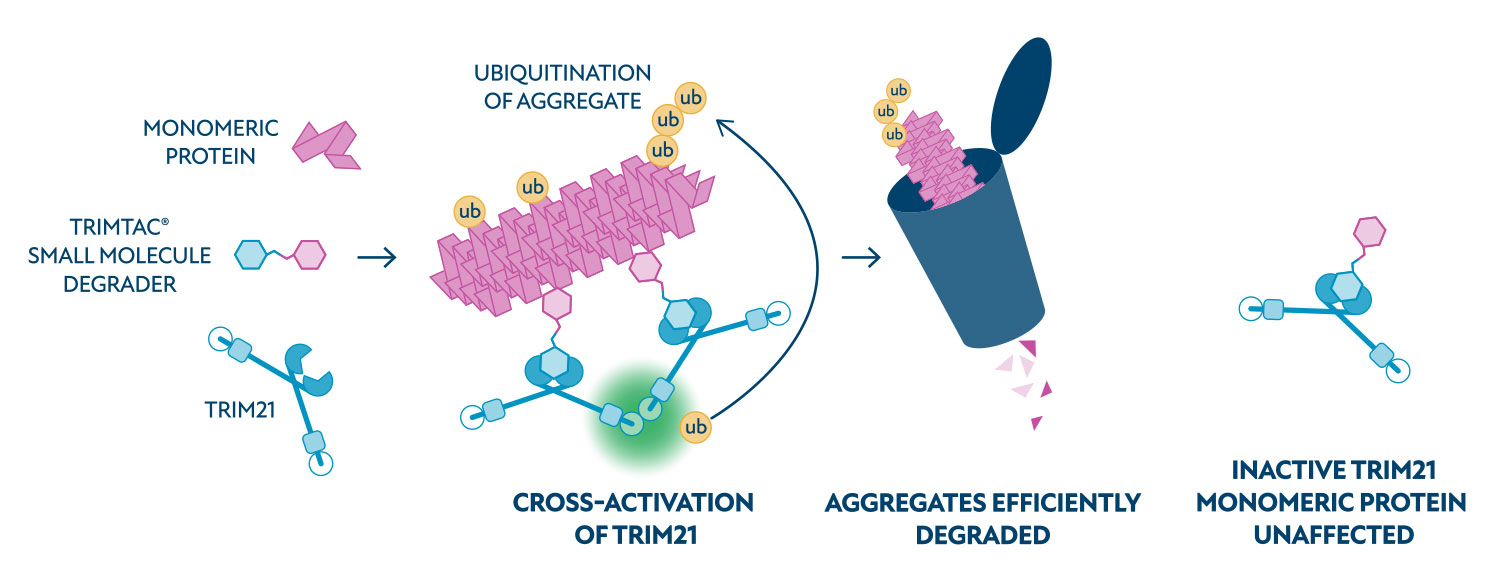
TRIM21 has a unique catalytic mechanism and only becomes activated by ‘substrate-induced clustering’. This means only multimeric targets such as oligomers, aggregates or functional assemblies can switch it on. Once activated, TRIM21 rapidly ubiquitinates its targets drivingdegradation of aggregates across the entire size spectrum – from small complexes and soluble oligomers, to large fibrillar structures. In the case of tau, the TRIM21 mechanism can also neutralise any aggregation seeding activity in treated cells.
As TRIM21 is activated exclusively by clustered targets, TRIMTECH does not need to engineer aggregate-selective ligands. These mechanistic features, coupled with tuneable aggregate selectivity, provide major strategic advantages over conventional degrader approaches.
TRIMTAC® and TRIMGLUE® – Proprietary small molecule degraders
TRIMTAC® and TRIMGLUE® molecules are designed to recruit TRIM21, an E3 ligase, to disease-causing aggregated protein targets, leading to their efficient degradation via the ubiquitin-proteasome system.
TRIMTAC®
TRIMTAC® molecules are bispecific ‘chimeric’ molecules that recruit TRIM21 to the target protein of interest.
TRIMGLUE®
TRIMGLUE® molecules bring a protein target and TRIM21 together by exploiting cryptic mutual binding interactions. TRIMGLUE® molecules can be applied where no suitable ligands are known for the target.
Programs
Our portfolio of programs are focused on Alzheimer’s, Parkinson’s, Huntington’s and other neurodegenerative and inflammatory disorders.
Publications by our Scientific Founders
James, L.C, et al; Structural basis for PRYSPRY-mediated tripartite motif (TRIM) protein function. Proceedings of the National Academy of Sciences of the United States of America, 2007, 104(15): p6200-5.
Mukadam, A.S., et al., Cytosolic antibody receptor TRIM21 is required for effective tau immunotherapy in mouse models. Science, 2023. 379(6639): p. 1336-1341.
Benn, J., et al., Aggregate-selective removal of pathological tau by clustering-activated degraders. Science, 2024. 385(6712): p. 1009-1016.
Miller, L.V.C., et al., Co-opting templated aggregation to degrade pathogenic tau assemblies and improve motor function. Cell, 2024. 187(21): p. 5967-5980 e17.
Mallery, D.L., et al., Antibodies mediate intracellular immunity through tripartite motif-containing 21 (TRIM21). Proc Natl Acad Sci U S A, 2010. 107(46): p. 19985-90.
McEwan, W.A., et al., Cytosolic Fc receptor TRIM21 inhibits seeded tau aggregation. Proc Natl Acad Sci U S A, 2017. 114(3): p. 574-579.
Zeng, J., et al., Target-induced clustering activates Trim-Away of pathogens and proteins. Nat Struct Mol Biol, 2021. 28(3): p. 278-289.
Clift, D., et al., A Method for the Acute and Rapid Degradation of Endogenous Proteins. Cell, 2017. 171(7): p. 1692-1706 e18.
Kiss, L., et al., Trim-Away ubiquitinates and degrades lysine-less and N-terminally acetylated substrates. Nat Commun, 2023. 14(1): p. 2160.
Hauler, F., et al., AAA ATPase p97/VCP is essential for TRIM21-mediated virus neutralization. Proceedings of the National Academy of Sciences of the United States of America, 2012. 109(48): p. 19733-8.